Blind Patients Gain Vision Thanks to Bionic Eye Breakthrough in Clinical Trials

- A second-generation bionic eye restored functional vision in four patients with inherited blindness from retinitis pigmentosa, allowing them to detect edges, shapes, movement, and navigate their environments with greater independence.
- The device proved remarkably durable, with 97% of electrodes still functioning 2.7 years after implantation and no complications reported, signaling strong potential for long-term use.
- Worldwide pivotal trials and a third-generation device are in development, with commercial release targeted for 2026, which could reshape how nurses care for patients with degenerative vision loss.

Results from the first clinical trial of Australia's second-generation bionic eye show the device delivered substantial improvements in functional vision, daily activities, and quality of life for four participants who were completely blind from retinitis pigmentosa.
The trial, led by the Centre for Eye Research Australia (CERA), Bionics Institute, University of Melbourne, and Royal Victorian Eye and Ear Hospital, tracked patients from their implant surgery in 2018 through 2021. Detailed outcomes were published in the journal Ophthalmology Science, and the findings are turning heads across the medical community.
The device has also been granted Breakthrough Device designation by the FDA, putting it on a fast track toward potential commercial availability as early as 2026.
How the Bionic Eye Works
The bionic eye system comprises three core components: an electrode array with 44 platinum disc electrodes surgically implanted behind the eye, a video camera mounted on a pair of glasses, and a processing unit that converts visual information into electrical signals.
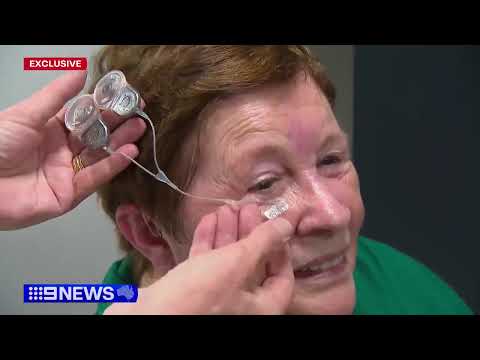
Professor James Fallon, Head of Research at the Bionics Institute, explained the mechanism: "The camera converts images into electrical pulses delivered by the electrode array that activate retinal cells and create flashes of light called phosphenes to help patients detect edges, shapes and movement."
Unlike gene therapies or stem cell treatments that attempt to repair damaged retinal tissue, this prosthetic approach bypasses the damaged photoreceptor cells entirely and directly stimulates the remaining viable neurons in the retina. Retinitis pigmentosa, the condition affecting all four trial participants, is an inherited disease that progressively destroys the retina's light-sensing cells and affects roughly 1 in 4,000 people in the United States.
>>Listen to The Latest Nurse News Podcast
Trial Results Show Real-World Impact
Associate Professor Penny Allen, the trial's Principal Investigator and a vitreo-retinal surgeon at the Royal Victorian Eye and Ear Hospital, led the clinical assessment of all four participants. The results were striking.
Participants demonstrated enhanced navigation and obstacle avoidance in both clinical settings and everyday environments. They could locate doorways, find items on tabletops, and move through unfamiliar spaces with significantly greater confidence. Several participants reported being able to detect people at train stations and locate their spouses in cafes, activities that had been impossible without the device.
The technology supplemented existing mobility aids like canes and guide dogs rather than replacing them, giving users an additional layer of environmental awareness that reduced their need for human assistance during community outings and local travel.
Perhaps most importantly, the device proved durable and safe over the long term. The electrode array remained stable behind the retina without complications, and 97% of electrodes were still functioning at the 2.7-year mark.
What Comes Next: Generation 3 and Global Trials
Dr. Ash Attia, CEO of Bionic Vision Technologies, the Australian medical technology company commercializing the device, confirmed that a third-generation bionic eye is now in development. The next-generation system will employ an innovative Neural Activity Shaping (NAS) stimulation strategy designed to further improve the quality of restored vision.
The company is preparing for worldwide pivotal trials across the United States, Europe, and Australia ahead of seeking regulatory approval in key markets. With the FDA Breakthrough Device designation already in hand, the path to approval could move faster than for a standard medical device.
Meanwhile, researchers at the Bionics Institute are also exploring hybrid stimulation techniques that combine electrical signals with other approaches. Professor Rachael Richardson noted that "bionic eyes currently in clinical trials are predominantly electrical-only devices" and that hybrid stimulation could be "a missing piece which greatly improves the level of vision restored."
What Nurses Need to Know
For nurses working in ophthalmology, neurology, surgical recovery, or community health, this technology represents a shift in how degenerative vision loss may be managed in the near future. As retinal prostheses move closer to commercial availability, nurses will be on the front lines of patient education, pre-operative preparation, post-surgical monitoring, and long-term rehabilitation support.
Patients receiving bionic eye implants require months of visual training after surgery to learn how to interpret the phosphene patterns the device produces. This rehabilitation process is similar to what cochlear implant recipients undergo, and nurses will play a critical role in helping patients set realistic expectations and stay engaged through the training period.
Post-operative monitoring is also key. While this trial reported no complications, other retinal prosthesis trials have documented side effects including ocular hypertension, retinal tears, and subretinal hemorrhage. Nurses should be prepared to assess for these complications and coordinate with ophthalmology teams accordingly.
With roughly 100,000 Americans living with retinitis pigmentosa and millions more affected by other forms of degenerative blindness, the potential patient population is significant. Nurses in primary care and community settings should also be aware of this emerging technology so they can guide patients and families toward appropriate specialists and clinical trial opportunities. Those interested in participating in future trials can check the Centre for Eye Research Australia website for enrollment information.
🤔 Nurses, have you cared for patients with retinitis pigmentosa or other forms of progressive vision loss? How do you think bionic eye technology could change the way you support these patients? Share your thoughts in the comments!
If you have a nursing news story that deserves to be heard, we want to amplify it to our massive community of millions of nurses! Get your story in front of Nurse.org Editors now - click here to fill out our quick submission form today!